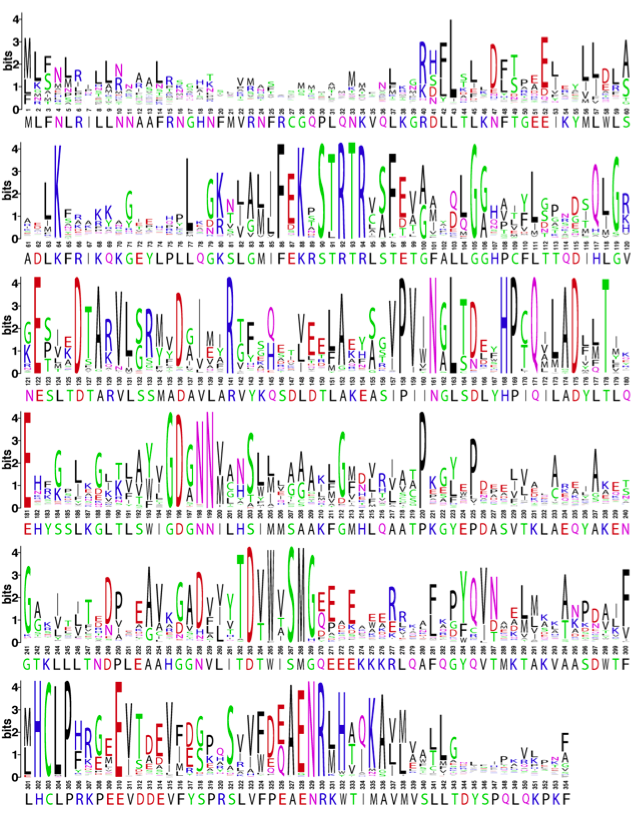
OTCase is a highly conserved enzyme. Sequences from bacteria to man all align readily, showing strong conservation of residues involved in binding carbamyl phosphate and ornithine. The severity of OTC deficiency due to single amino acid changes (missense mutation) in part is due to the differences in physical and chemical properties of the amino acid substitution, but also is due to the mutation location. Amino acids that are important for the three dimensional fold of OTCase or important for binding substrates or catalyzing the enyzmatic reaction are very highly conserved. A collorary is that regions of OTCase that are highly variable amongst organisms also appear to be regions where mutations tend to result in a milder form of OTC deficiency.
Shown below is a logo representation of an alignment of 566 OTCases. The numbering corresponds to the human sequence by removing insertions relative to the human sequence. The size of the letters indicates the degree of sequence conservation and most of these have catalytically or structurally important roles. The human sequence is shown below the number track. A scalable PDF version of this image is available here.